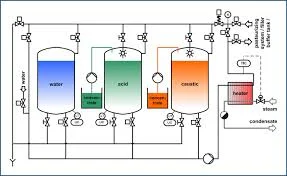
Cleaning-in-Place (CIP) Systems
5 products available
What are Cleaning-in-Place (CIP) Systems?
Cleaning-in-Place (CIP) systems are automated systems designed for the efficient cleaning of process equipment without the need for disassembly. This technology allows for the cleaning of pipelines, tanks, and other components in industries such as food and beverage, pharmaceuticals, and chemicals. CIP systems typically utilize a combination of water, cleaning agents, and sanitizing solutions that are circulated through the equipment to ensure thorough cleaning and sanitation.
The primary uses of CIP systems include maintaining hygiene standards, reducing downtime, and enhancing operational efficiency. By automating the cleaning process, businesses can save time and labor costs while ensuring that their equipment meets regulatory and quality standards. CIP systems are particularly valuable in industries where cleanliness is critical, as they help prevent contamination and ensure product safety.
The relevance of CIP systems in the market is growing, driven by increasing regulatory requirements and consumer demands for quality. As industries expand and evolve, the need for effective cleaning solutions becomes more pronounced. Companies are investing in advanced CIP technologies to streamline their operations and ensure compliance with safety standards.
𝗞𝗲𝘆 𝗙𝗲𝗮𝘁𝘂𝗿𝗲𝘀 𝗮𝗻𝗱 𝗦𝗽𝗲𝗰𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
CIP systems have several key features that enhance their effectiveness and usability. Understanding these specifications is essential for selecting the right system for your business needs.
Key specifications include:
1. Cleaning Cycle Time
- The duration for a complete cleaning cycle.
- Typically ranges from 30 minutes to several hours depending on system design.
2. Chemical Compatibility
- The ability of the CIP system to handle various cleaning agents.
- Must be compatible with acids, alkalis, and detergents commonly used in the industry.
3. Flow Rate
- The volume of cleaning solution that can be circulated per minute.
- Usually ranges from 10 to 1000 liters per minute based on system size and design.
4. Temperature Control
- The capacity to maintain specific temperatures for effective cleaning.
- Often operates within a range of 60 to 85 degrees Celsius.
5. Automation Level
- The degree to which the cleaning process is automated.
- Can range from fully automated systems to semi-automated setups.
6. Monitoring Features
- Availability of real-time monitoring for cleaning parameters.
- Systems may include sensors for tracking flow rate, temperature, and chemical concentration.
7. System Materials
- The materials used in the construction of CIP systems.
- Typically made from stainless steel for corrosion resistance and hygiene.
8. Compliance Standards
- Adherence to industry standards such as CE marking and ISO certifications.
- Ensures that the system meets safety and performance requirements.
In summary, understanding the key features and specifications of CIP systems is crucial for making informed purchasing decisions. These specifications ensure that the systems are efficient, safe, and suitable for the specific cleaning needs of various industries.
𝗖𝗼𝗺𝗺𝗼𝗻 𝗔𝗽𝗽𝗹𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀 𝗮𝗻𝗱 𝗨𝘀𝗲 𝗖𝗮𝘀𝗲𝘀
CIP systems are utilized across a variety of industries where cleanliness and hygiene are paramount.
1. Food and Beverage Industry: CIP systems are critical in breweries, dairies, and food processing plants to ensure that equipment is sanitized and free from contaminants, thus maintaining product safety.
2. Pharmaceutical Industry: In pharmaceutical manufacturing, CIP systems are essential for cleaning equipment used in drug production, ensuring compliance with strict health regulations.
3. Chemical Manufacturing: CIP systems help in maintaining the cleanliness of equipment used in the production of chemicals, preventing cross-contamination between different batches.
4. Biotechnology: In biotech facilities, CIP systems are used to clean bioreactors and fermentation tanks to ensure that the production environment remains sterile.
5. Dairy Processing: Dairy plants utilize CIP systems to clean milking equipment, storage tanks, and processing lines to prevent bacterial growth and spoilage.
6. Cosmetics: In the cosmetics industry, CIP systems ensure that mixing and filling equipment is cleaned thoroughly to maintain product integrity and safety.
7. Beverage Production: Beverage producers rely on CIP systems to keep bottling and canning lines free from contaminants, ensuring high-quality products.
𝗣𝗿𝗼𝗱𝘂𝗰𝘁 𝗩𝗮𝗿𝗶𝗮𝗻𝘁𝘀 𝗮𝗻𝗱 𝗦𝘂𝗯𝗰𝗮𝘁𝗲𝗴𝗼𝗿𝗶𝗲𝘀
CIP systems come in various configurations to suit different industrial needs. Although specific subcategories are not available, the diversity in design allows businesses to choose systems based on their unique requirements.
Standard CIP System
This variant is designed for general cleaning purposes across various industries. It features programmable cleaning cycles and can handle a variety of cleaning agents.
Compact CIP System
Ideal for smaller operations, this version occupies less space while still providing effective cleaning capabilities. It is perfect for businesses with limited floor space.
Mobile CIP System
This variant is designed for flexibility and can be moved between different cleaning locations. It is suitable for industries requiring cleaning in multiple areas or facilities.
𝗕𝗲𝗻𝗲𝗳𝗶𝘁𝘀 𝗮𝗻𝗱 𝗔𝗱𝘃𝗮𝗻𝘁𝗮𝗴𝗲𝘀
Investing in Cleaning-in-Place systems offers numerous advantages for businesses.
Key benefits include:
1. Time Efficiency: CIP systems significantly reduce cleaning time compared to manual methods, allowing for quicker turnaround between production runs.
2. Labor Savings: Automation reduces the need for manual labor, freeing up staff for other critical tasks and minimizing labor costs.
3. Consistent Results: Automated cleaning ensures uniform application of cleaning agents, leading to consistent cleaning results and higher quality standards.
4. Compliance Assurance: CIP systems help businesses meet stringent regulatory requirements by providing documentation and traceability of cleaning processes.
5. Reduced Water Usage: CIP systems optimize water use through recycling features, contributing to sustainability efforts and reducing utility costs.
6. Enhanced Safety: By minimizing manual handling of chemicals, CIP systems improve workplace safety and reduce the risk of accidents.
𝗕𝘂𝘆𝗶𝗻𝗴 𝗚𝘂𝗶𝗱𝗲 𝗳𝗼𝗿 𝗕𝟮𝗕 𝗕𝘂𝘆𝗲𝗿𝘀
When sourcing Cleaning-in-Place (CIP) systems, several factors should be considered to ensure you select the right solution for your business.
1. Quality Assessment: Evaluate the quality of the CIP system by reviewing material certifications and performance standards. Look for systems that meet industry requirements and have a track record of reliability.
2. Supplier Verification: Vet suppliers by checking their credentials, customer reviews, and industry expertise. A reliable supplier can provide support and maintenance services post-purchase.
3. Pricing and MOQ: Understand the pricing structure and minimum order quantities. Consider the total cost of ownership, including installation, maintenance, and operational costs.
4. Delivery and Lead Times: Inquire about delivery timelines and lead times to ensure that the system can be integrated into your operations as planned.
5. Compliance Requirements: Ensure that the CIP system complies with relevant industry regulations and safety standards. This is crucial for maintaining product integrity and market access.
𝗖𝗼𝗺𝗽𝗹𝗶𝗮𝗻𝗰𝗲 𝗮𝗻𝗱 𝗖𝗲𝗿𝘁𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
Compliance and certifications are critical aspects of Cleaning-in-Place systems. They ensure that the systems meet established safety and performance standards.
Relevant quality standards include ISO 9001, which focuses on quality management systems, and HACCP (Hazard Analysis Critical Control Point), which is essential for food safety. These standards help businesses implement effective quality control measures.
Industry certifications, such as CE marking, indicate that the CIP system complies with European health, safety, and environmental protection standards. This certification is vital for gaining access to markets in the European Union.
Safety and testing requirements for CIP systems are stringent, as they must ensure that the equipment operates safely under various conditions. Regular testing and maintenance are essential to uphold these standards.
Regional compliance considerations may vary, so businesses should be aware of local regulations that impact CIP system operation, especially in international markets.
Why Source Cleaning-in-Place (CIP) Systems from Pepagora?
Pepagora provides a trusted platform for sourcing Cleaning-in-Place (CIP) systems, offering several key advantages for businesses.
Verified Supplier Network: Pepagora ensures that all suppliers are thoroughly vetted for quality and reliability. This network allows businesses to confidently source systems that meet their specific requirements.
Streamlined RFQ Process: The request for quotation (RFQ) process is designed to be user-friendly, enabling buyers to quickly receive and compare quotes from multiple suppliers. This efficiency helps in making informed purchasing decisions.
Trade Assurance and Support: Pepagora offers trade assurance, providing buyers with protection against potential issues during the transaction. Additionally, support services are available to assist with any inquiries or challenges that may arise.
Regional Coverage: Pepagora's reach extends across India, the GCC, and Southeast Asia, ensuring that businesses in these regions can access high-quality CIP systems tailored to their local market needs.
Newly Added
Recently Added
Massreach Associates
Professional Harvest Operations And Vegetable Post Harvest Management Services
Beverage Manufacturing Carbonated Soft Drinks Plants High Efficiency
Premium Packaged Drinking Water Plants For Industries
Anmol Pharma Equipments
Industrial Conveyors For Material Handling: Durable & Efficient
Automated CIP Vessel Systems For Tanks, Mixers, Blenders & IBC Bins
Industrial Filling Vessels For Precise Liquid Dispensing In Pharma
Armaan Industries
Premium PVC Shrink Sleeves For Product Labeling And Tamper Evidence
Premium PVC Shrink Labels For PET Bottles Durable Custom Branding
Professional PVC Shrink Plain Films 20Mic 80Mic Durable Packaging
Sri Koputaiya Nayaki Industries
Commercial Dough Mixer Machine For Bakery And Food Production
Wet Garlic Peeling Machine For Food Preparation Facilities
Automatic Garlic Peeler Machine For Bulk Processing Units
Why Businesses Trust Pepagora
Thousands of companies trust Pepagora to source smarter and sell faster
Expand Your Sales Globally
25000+Products listed.Get discovered by global buyers
Tools to boost visibility
Build your own catalog

Source Smarter,Grow Faster
4000+Active RFQs
100+ Countries served
Easy Sourcing Tools