Similar Products
Description
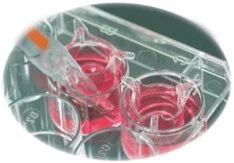
Thin Cert cell culture inserts represent advanced laboratory equipment designed for precise in vitro cell culture applications. These inserts feature stable clear polystyrene housi...
Thin Cert cell culture inserts represent advanced laboratory equipment designed for precise in vitro cell culture applications. These inserts feature stable clear polystyrene housing with a sealed PET capillary pore membrane, ensuring optimal visibility and barrier integrity during experiments. The unique hanging geometry and self-lift design simplify pipetting operations while maintaining sterile conditions through single blister packing. Physical surface treatment enhances cell adhesion properties, making these inserts reliable for critical research applications where consistency and reproducibility are paramount. Researchers benefit from improved gas exchange capabilities and enhanced pipetting access, reducing experimental variability and supporting accurate data generation across multiple trial runs. The product's construction ensures reliable performance in various laboratory environments while maintaining cell viability and experimental integrity throughout extended study periods. These cell culture inserts serve vital roles across multiple industries including pharmaceutical development, biomedical research, and academic institutions. Pharmaceutical companies utilize them for drug permeability testing and toxicology screening during preclinical development phases. Biotechnology firms employ these inserts for stem cell research, cancer studies, and tissue engineering applications. Academic research laboratories rely on them for cell migration assays, co-culture experiments, and barrier function studies. The inserts' compatibility with standard multiwell plates makes them suitable for contract research organizations conducting high-throughput screening services. Clinical diagnostics laboratories also use these products for developing diagnostic assays and studying disease mechanisms at cellular levels. The versatility of these inserts makes them valuable across multiple research disciplines and experimental requirements. Businesses choosing Thin Cert inserts gain significant value through enhanced research reliability and operational efficiency. The product's consistent quality ensures reproducible results across experiments, reducing costly repeats and accelerating research timelines. The sterile blister packaging minimizes contamination risks while simplifying inventory management and storage. Manufacturers maintain strict quality control standards, delivering products that meet international laboratory requirements and compliance standards. This reliability translates into reduced downtime and higher throughput for research facilities, ultimately supporting faster innovation and development cycles for organizations investing in cellular research technologies. The product's design features contribute to time savings during experimental setup and execution, allowing researchers to focus on data analysis rather than equipment concerns. Key Features: - Stable clear polystyrene housing for optimal visibility during experiments - Hanging geometry design for secure placement in multiwell plates - Sealed PET capillary pore membrane ensuring reliable barrier function - Single sterile blister packing maintaining product integrity until use - Physical surface treatment enhancing cell adhesion capabilities Benefits: - Consistent experimental results through reliable barrier integrity - Reduced contamination risk with sterile single-use packaging - Enhanced research efficiency with simplified pipetting access - Improved cell adhesion for more accurate study outcomes - Compatibility with standard laboratory equipment and protocols
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Thin Cert cell culture inserts are essential tools for biomedical research and pharmaceutical development, enabling precise cell growth studies in controlled environments. Their design supports advanced cell migration, co-culture experiments, and barrier function assessments with high reproducibility across diverse laboratory settings. These inserts maintain consistent performance for critical research applications where accuracy and reliability are paramount requirements for successful experimental outcomes. These inserts are widely adopted in academic institutions, contract research organizations, and biotech companies for toxicology screening, drug permeability testing, and cancer research. The standardized format ensures compatibility with multiwell plates, making them suitable for high-throughput screening and routine cell culture applications requiring reliable performance. Laboratories benefit from the consistent quality and ease of use that these inserts provide across various research protocols and experimental setups. - Drug discovery and development processes for permeability and absorption studies - Cancer research involving tumor cell migration and invasion assays - Toxicology screening to evaluate compound effects on cell viability - Stem cell research and differentiation monitoring in controlled environments - Biomedical studies of epithelial and endothelial barrier function models
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Bio Globus, New Delhi
Distributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Lab Cell Culture Inserts Thin Cert Polystyrene PET Membrane Sterile
Thin Cert cell culture inserts provide reliable in vitro cell growth with clear polystyrene housing, PET membrane, and sterile blister packing. Ideal for labs requiring precision, improved adhesion, and simplified pipetting access for consistent research results.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Bio Globus
Distributor / Wholesaler